Tidda Herz
Die Selbstmanagement-App für Patientinnen und Patienten mit Herzinsuffizienz in Deutschland
Über Tidda Herz
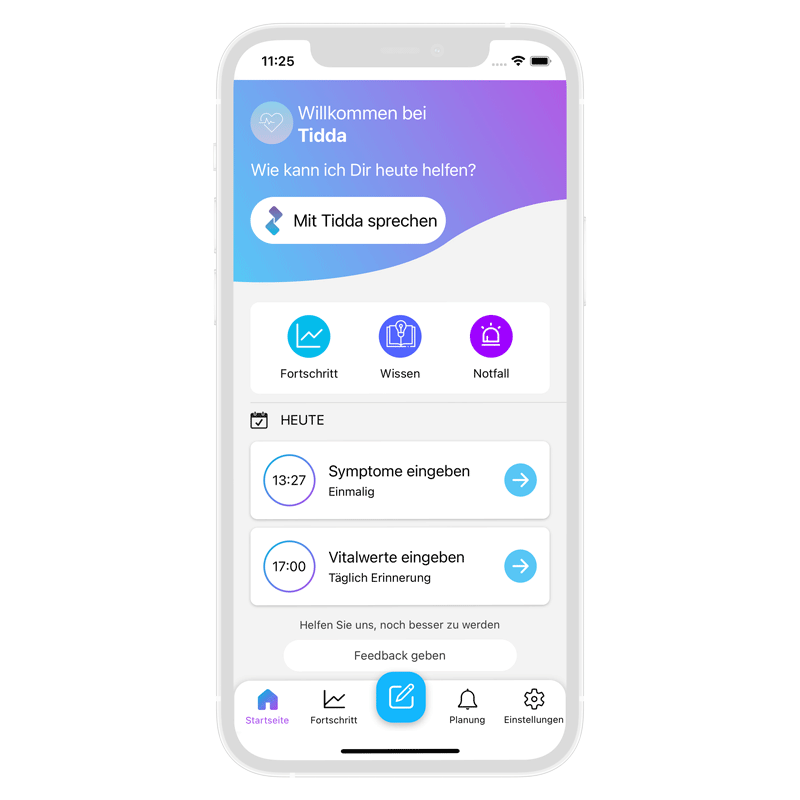
Tidda Herz unterstützt Sie beim Umgang mit Herzinsuffizienz im Alltag. Messen Sie Ihre Vitalwerte und reagieren Sie rechtzeitig auf Verschlechterungen. Tidda wurde von Kardiologen trainiert, um Ihre Fragen zur Krankheit im Dialog zu beantworten. Als App in Ihrem Smartphone ist Tidda immer für Sie da.
Ich bin Tidda
Demo-Video abspielen
Auszeichnungen
Datenschutz und Datensicherung
Die App verarbeitet persönliche Daten und
Gesundheitsdaten, welche getrennt voneinander
gespeichert werden.
Persönliche Daten helfen uns, Sie sicher zu identifizieren. Das ist wichtig, damit nur Sie selbst Zugang zu Ihren Gesundheitsdaten bekommen. Zu den persönlichen Daten gehören: Name, Telefonnummer, E-Mail. Wenn Sie die App installieren, wird zudem für Sie automatisch eine individuelle Identifikationsnummer ("ID") generiert.
Gesundheitsdaten unterstützen Sie, Ihre Krankheit im Alltag selbst zu managen. Gesundheitsdaten sind Symptome und Vitalwerte, die Sie eingeben, Medikamente, die Sie aktuell einnehmen und Erinnerungen, die Sie selbst setzen.
Persönliche Daten helfen uns, Sie sicher zu identifizieren. Das ist wichtig, damit nur Sie selbst Zugang zu Ihren Gesundheitsdaten bekommen. Zu den persönlichen Daten gehören: Name, Telefonnummer, E-Mail. Wenn Sie die App installieren, wird zudem für Sie automatisch eine individuelle Identifikationsnummer ("ID") generiert.
Gesundheitsdaten unterstützen Sie, Ihre Krankheit im Alltag selbst zu managen. Gesundheitsdaten sind Symptome und Vitalwerte, die Sie eingeben, Medikamente, die Sie aktuell einnehmen und Erinnerungen, die Sie selbst setzen.
Beim Umgang mit Ihren Daten halten wir uns streng an die
allgemeinen Datenschutzbestimmungen der EU sowie die
lokalen Datenschutzgesetze. Insbesondere werden Ihre
persönlichen Daten nicht ohne Ihr Einverständnis an
Dritte weitergegeben.
Ihre persönlichen Daten werden auf folgende Weise von Gesundheitsdaten getrennt:
Ihre persönlichen Daten werden auf folgende Weise von Gesundheitsdaten getrennt:
- Persönliche Daten und Gesundheitsdaten werden in zwei verschiedenen Datenbanken gespeichert.
- Die Verknüpfung zwischen den beiden Datenbanken ist nur über die eindeutige individuelle Identifikationsnummer ("ID") möglich.
- Daten, die einen einzelnen Patienten potenziell identifizieren könnten, werden nicht in den Gesundheitsdaten gespeichert.
Während Ihrer Dateneingabe oder Erhebung in der mobilen
App werden die Daten auf Ihrem Smartphone verschlüsselt
gespeichert.
Sollten Sie eine Datenübertragung an Ihren behandelnden Arzt wünschen, erfolgt diese innerhalb der Plattform bis zu dem Monitoring Dashboard für Ihren Arzt in verschlüsselter Form durch eine Standard-Sicherheitstechnologie (Secure Sockets Layer). Dies ermöglicht eine verschlüsselte Verbindung zwischen zwei Computern und damit die sichere Übertragung sensibler Informationen.
Alle Daten, die in die Plattform übertragen werden, sind verschlüsselt, so dass es unmöglich ist, dass die Kommunikationsdaten von externen Sicherheitsbedrohungen ausgenutzt werden können.
Die anonymisierten Daten werden auf einem vom Technologieanbieter Zana Technologies GmbH zur Verfügung gestellten Server in Deutschland gespeichert, der den höchsten Sicherheitsstandards entspricht
Sollten Sie eine Datenübertragung an Ihren behandelnden Arzt wünschen, erfolgt diese innerhalb der Plattform bis zu dem Monitoring Dashboard für Ihren Arzt in verschlüsselter Form durch eine Standard-Sicherheitstechnologie (Secure Sockets Layer). Dies ermöglicht eine verschlüsselte Verbindung zwischen zwei Computern und damit die sichere Übertragung sensibler Informationen.
Alle Daten, die in die Plattform übertragen werden, sind verschlüsselt, so dass es unmöglich ist, dass die Kommunikationsdaten von externen Sicherheitsbedrohungen ausgenutzt werden können.
Die anonymisierten Daten werden auf einem vom Technologieanbieter Zana Technologies GmbH zur Verfügung gestellten Server in Deutschland gespeichert, der den höchsten Sicherheitsstandards entspricht